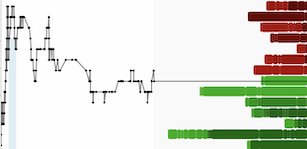
- Largest head-to-head randomized clinical trial between DTG/3TC and BIC/FTC/TAF, conducted by SEIMC-GeSIDA Foundation (FSG) showed DTG/3TC demonstrated non-inferior efficacy compared to BIC/FTC/TAF as a switch regimen for virologically-suppressed adults living with HIV over 48 weeks of therapy.
- DTG/3TC-treated individuals had significantly less weight gain compared to those randomized to BIC/FTC/TAF.
- In 2022, the Canadian HIV incidence rate grew by 15% in comparison to 2020, a total of 1,848 new cases. 1
- According to the latest surveyed data, there is an estimated 65,270 Canadians living with HIV. 1
MONTREAL, July 31, 2024 /CNW/ - ViiV Healthcare, the global specialist HIV company majority owned by GSK, with Pfizer and Shionogi as shareholders, announced the 48-week findings from PASO DOBLE (GeSIDA 11720 study), the largest head-to-head, phase IV randomized clinical trial (RCT) investigating the 2-drug regimen DOVATO (dolutegravir/lamivudine [DTG/[3TC]) compared to the 3-drug regimen BIKTARVY (bictegravir/emtricitabine]/tenofovir alafenamide fumarate [BIC/FTC/TAF]) for the treatment of HIV-1 in people who are virologically suppressed and who could benefit from treatment optimization.2
Findings showed that switching to DTG/3TC in virologically suppressed adults living with HIV demonstrated non-inferior efficacy in maintaining viral suppression compared with switching to BIC/FTC/TAF.3 This data was recently presented at the 25th International AIDS Conference (AIDS 2024), in Munich, Germany.
Harmony P. Garges, M.D., MPH, Chief Medical Officer at ViiV Healthcare, said: "The results from PASO DOBLE show that Dovato demonstrated non-inferior efficacy compared to Biktarvy, and that the average weight gain for trial participants taking DTG/3TC was significantly lower than those taking BIC/FTC/TAF over the course of the year. This is a meaningful outcome, as treatment-related weight gain is an important topic for many people living with HIV. At ViiV Healthcare we're dedicated to bringing innovative HIV treatments to people living with HIV that are not only safe and effective, but also address their specific needs beyond viral suppression."
Jean-Francois Fortin, Country Medical Director, Canada at ViiV Healthcare, said: "With multiple options available for the treatment of HIV, it's our responsibility to further study and monitor available therapies to ensure that Canadians living with HIV are receiving the best possible care. We are excited to see that the results from PASO DOBLE show that Dovato, a 2-drug regimen, offers comparable efficacy, as well as less weight gain compared to BIC/FTC/TAF through 48 weeks. At ViiV Healthcare Canada, our commitment is to provide HIV treatments that address the various needs of people living with HIV, which begins with obtaining viral suppression."
In the PASO DOBLE clinical trial, 553 people living with HIV and virally suppressed switched treatment to either DTG/3TC (n=277) or BIC/FTC/TAF (n=276). The study population included individuals who were on therapy that could be optimized, such as multiple tablet regimens, or those containing pharmacokinetic boosting agents or drugs with cumulative toxicity, such as efavirenz or tenofovir disoproxil fumarate (TDF).3 The study met its primary endpoint when DTG/3TC demonstrated non-inferior efficacy versus BIC/FTC/TAF based on the proportion of participants with viral RNA ≥50 copies/mL at 48 weeks using the FDA snapshot and a 4% non-inferiority margin in the exposed intention-to-treat population.3
At 48 weeks, DTG/3TC was non-inferior to BIC/FTC/TAF (risk difference between DTG/3TC [2.2%] minus BIC/FTC/TAF [0.7%] of 1.4%, 95% CI -0.5 to 3.4). One participant in the BIC/FTC/TAF arm and zero in the DTG/3TC arm had protocol-defined confirmed virological failure through week 48 (HIV-1 RNA ≥50 c/mL followed by a second consecutive HIV-1 RNA assessment ≥200 c/mL).3
The study found that weight increased significantly more in participants who switched to BIC/FTC/TAF (adjusted mean change 1.81kg, 95% CI 1.28-2.34) than in those who switched to DTG/3TC (adjusted mean change 0.89kg, 95% CI 0.37-1.41) [difference 0.92kg, 95% CI 0.17-1.66] through week 48. Equally, the proportion of participants with weight gain greater than 5% at week 48 was significantly higher at 29.9% for BIC/FTC/TAF compared to 20% for DTG/3TC (adjusted OR 1.81, 95% CI 1.19-2.76).3
Weight change with DTG/3TC did not differ between men and women or based on the previous regimen of participants, whereas the proportion of trial participants experiencing greater than 5% weight gain with BIC/FTC/TAF was approximately 45% higher than those taking DTG/3TC when switching from a regimen with abacavir (30.6% BIC/FTC/TAF vs 21.1% DTG/3TC), and about 2-fold higher when switching from a regimen with TDF (40.7% BIC/FTC/TAF vs 19.5% DTG/3TC). Safety was comparable through week 48 and consistent with known safety profiles. There were few discontinuations due to adverse events in both study arms (DTG/3TC = 1, 0.4%; BIC/FTC/TAF = 2, 0.7%), with no differences between arms.3
About PASO DOBLE
The PASO DOBLE (NCT04884139) randomized clinical trial is a phase IV, open-label, randomized multicentre clinical trial evaluating the efficacy of DTG/3TC versus BIC/FTC/TAF for the maintenance of virologic suppression in people living with HIV-1, conducted in 30 sites across Spain. Virologically suppressed people living with HIV on regimens containing ≥1 pill/day, boosters, or drugs with cumulative toxicity such as efavirenz or TDF were eligible and were randomized (1:1) to switch to either DTG/3TC or BIC/FTC/TAF. The primary endpoint was the proportion of people living with HIV with RNA ≥50 copies/mL at 48 weeks (FDA snapshot, 4% non-inferiority margin) in the intention-to-treat exposed population. Secondary outcomes measured included, among others, absolute weight gain, BMI change, and the proportion of participants with weight change greater than 5%.3
About DOVATO
DOVATO is indicated as a complete regimen for the treatment of Human Immunodeficiency Virus type 1 (HIV-1) infection in adults and adolescents 12 years of age and older and weighing at least 40 kg.3
DOVATO is approved in Canada, US, Europe, Japan, Australia, and other countries worldwide.
Please consult the Product Monograph at www.viivhealthcare.ca for complete safety information. The Product Monograph is also available by calling 1-877-393-8448.
About SEIMC-GeSIDA Foundation (FSG)
The SEIMC-GeSIDA Foundation (FSG) was created to encourage, promote, and support scientific and technical research and development, training, and publication of findings in the field of clinical microbiology and infectious diseases and associated conditions. FSG was founded by investigators from the Spanish Society of Clinical Microbiology and Infectious Diseases as a tool to promote high-quality investigation in the field of HIV infection and other infectious diseases. The Foundation also aims to respond to the scientific concerns of the group's members.
FSG is composed of qualified professionals with experience in the field of clinical trials and multicenter studies. Its streamlined infrastructure facilitates performance of clinical studies and responds to the needs of investigators in terms of methodology/statistical analysis and of logistics and management of trials and other multicenter studies. FSG also provides staff to run events such as scientific meetings and conferences (national and international) and to organize courses, lectures, talks, seminars, round-table talks, and specialized workshops.
For more information on the FSG, please visit https://fundacionseimcgesida.org/en/quienes-somos/
About HIV
HIV (human immunodeficiency virus) is a virus that attacks the body's immune system.4 HIV is spread by contact with certain bodily fluids of a person with HIV, most commonly during unprotected sex (sex without a condom or HIV medicine to prevent or treat HIV), or through sharing injected drug equipment.5
If HIV is not treated, it can lead to AIDS (acquired immunodeficiency virus). AIDS is the most severe stage of HIV (Stage 3).5 There is currently no cure for HIV, but with proper treatment and care, people with HIV can maintain a high quality of life and avoid passing HIV to others.
At the end of 2022, 39 million people lived with HIV around the world, with 1.3 million new diagnoses that year and 630,000 deaths from AIDS-related illnesses.6
About ViiV Healthcare
ViiV Healthcare is a global specialist HIV company established in November 2009 by GSK (LSE: GSK) and Pfizer (NYSE: PFE) dedicated to delivering advances in treatment and care for people living with HIV and for people who are at risk of acquiring HIV. Shionogi became a ViiV shareholder in October 2012. The company's aims are to take a deeper and broader interest in HIV and AIDS than any company has done before and take a new approach to deliver effective and innovative medicines for HIV treatment and prevention, as well as support communities affected by HIV.
For more information on the company, its management, portfolio, pipeline, and commitment, please visit www.viivhealthcare.ca.
_______________________________ |
1 Government of Canada. Canada's progress towards ending the HIV epidemic. Sourced from https://www.canada.ca/en/public-health/services/publications/diseases-conditions/canada-progress-towards-ending-hiv-epidemic.html. July 2024. |
2 P. Ryan, et al. Non-inferior efficacy and less weight gain when switching to DTG/3TC than when switching to BIC/FTC/TAF in virologically suppressed people with HIV (PWH): the PASODOBLE (GeSIDA 11720) randomised clinical trial. Presented at the 25th International AIDS Conference. July 2024 |
3 DOVATO Product Monograph (2023). Available at: www.viivhealthcare.ca |
4 CDC. About HIV. Available at: https://www.cdc.gov/hiv/about/?CDC_AAref_Val=https://www.cdc.gov/hiv/basics/whatishiv.html |
5 HIV.gov. What Are HIV and AIDS? Available at: https://www.hiv.gov/hiv-basics/overview/about-hiv-and-aids/what-are-hiv-and-aids/. |
6 UNAIDS. Global HIV & AIDS Statistics – Fact sheet. Available at: https://www.unaids.org/en/resources/fact-sheet |
SOURCE ViiV Healthcare

![]() View original content to download multimedia: http://www.newswire.ca/en/releases/archive/July2024/31/c2941.html
View original content to download multimedia: http://www.newswire.ca/en/releases/archive/July2024/31/c2941.html